 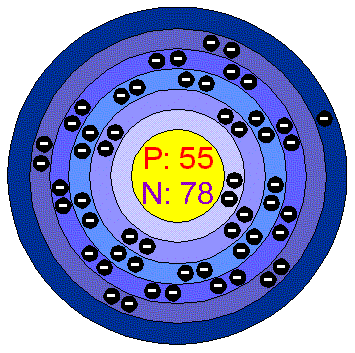
According to, cesium also reacts violently with sulfur, phosphorous, acids, and halogens (fluorine, chlorine, bromine, iodine and astatine). Because cesium reacts violently with water, it is treated as a hazardous material and is often kept under a layer of kerosene or mineral oil or in a vacuum to prevent it from reacting and igniting due to the moisture in the air.Cesium is highly reactive and combines readily with other elements, especially oxygen and other gases, and nonmetals, according to Encyclopedia Britannica.Cesium is the 50th most common element in Earth’s crust, according to PeriodicTable. According to Chemicool, the abundance of cesium in Earth's crust is about 3 parts per million by weight, and in the solar system 8 parts per billion by weight.Cesium is one of four metals that are liquid at or near room temperature the others are mercury (melting point of minus 37.9 F, or minus 38.8 C), gallium (melting point of 85.6 F, or 29.8 C) and francium (melting point of 80.6 F, or 27 C) according to Chemicool.Cesium-based atomic clocks lose one second per 100 million years. The official definition of a second is the time it takes for the cesium atom to vibrate 9,192,631,770 times between energy levels. Cesium is incredibly accurate at timekeeping and is used in atomic clocks.These metals are also very malleable, ductile and good heat and electrical conductors. Alkali metals are highly reactive, have one electron in their outer shell, and do not occur freely in nature, according to ChemicalElements.  
Cesium is an alkali metal, along with lithium, sodium, potassium, rubidium and francium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
